Identification and taxonomical evaluation of the germplasm resources
METHODOLOGY
Based on the lists of mulberry varieties that has been provided by the Aracne partners within the first year of the project (D1.4 – Progress report on the collected mulberry samples, see Table 1 and Suppl. Tables I-IV), selected trees from CREA, SCS Vratsa, UM and IMIDA germplasm collections were included in the monitoring of morphological descriptors and silica dried leaf material was send to CREA for molecular analyses.
High yielding sericultural and local varieties from the following collections were included:
- CREA Germplasm: The evaluation included 57 varieties from diverse taxonomical and geographical origins (Italy, China, France, India, Japan).
- Vratsa Germplasm: A total of 70 mulberry varieties were evaluated. The selection focused on local Bulgarian varieties and those from Eastern Europe and Central Asia (Armenia, Azerbaijan, Georgia, Romania, Ukraine, and Uzbekistan) to trace the possible spread of mulberries (and sericultural activity) from the East to the West. These varieties, introduced and planted at the SCS-Vratsa germplasm between 1965 and 2004, offer a unique insight into regional diversity.
- UM Germplasm: 166 distinct varieties were included in the analyses, representing 132 local historical white mulberry ( M. alba s.l.) trees inventoried in Slovenia (69) and in Hungary (63), along with 34 reference varieties used in sericulture and fruit processing. The varieties were planted mainly between years 2015-2018.
- IMIDA germplasm: 35 varieties varieties originating from Italy, Spain and Japan were included in the analysis.
The main issue was that most varieties are labelled with old traditional taxonomic names, synonyms (syn.), that apply to a taxon that now goes by a different scientific name. Moreover, most modern sericultural varieties originate from simple or complex spontaneous and/or artificial crosses between M. alba, M. indica and M. rubra. Therefore, most of the varieties belonging to M. alba and M. indica have been concurrently defined as sensu lato (s.l.), which means “in the broad sense”, whereas M. rubra varieties have been concurrently asigned as confer (cf.). This qualifier indicates that most of the diagnostic characters correspond to M. rubra, but some characters are unclear. The identification is provisional but is likely to be definitive after comparing with reference material. Varieties that are noted as M. rubra cf. could originate from simple or complex spontaneous and/or artificial crosses involving M. alba. Their genetic relationship is going to be analysed under the scope of ARACNE mulberry research. The collections also include forms (f.) of M. alba (M. alba f. pendula, M. alba f. pyramidalis), which denote a taxonomic category bellow the species level and refer to specific morphological differences. Variety (var.) is a taxonomic category that ranks between the subspecies and forma level, its members differing from others in minor but permanent or heritable characteristics.
According to the currently accepted classification, the varieties identified, evaluated, and sampled in the germplasm were taxonomically organised as shown in Table 1, taking known synonyms into account.
Table 1 - Current taxonomical status of M. alba s.l., M. indica s.l. and M. rubra cf. along with synonyms.
|
Current taxonomical status |
synonyms |
|
M. alba s.l. |
M. atropurpurea, M. alba var. atropurpurea |
|
M. alba f. pendula |
M. pendula |
|
M. pyramidalis |
|
|
M. indica |
M. australis |
|
|
M. bombycis M. kagayamae M. latifolia |
|
|
M. multicaulis, M. indica var. multicaulis M. planifolia, M. multicaulis var. planifolia M. rotundiloba |
|
M. rubra cf. |
Varieties that are noted as M. rubra cf. could originate from simple or complex spontaneous and/or artificial crosses involving M. alba. Their genetic relationship is going to be analysed under the scope of ARACNE mulberry research. |
Methodology for maintenance of mulberry accessions
Conservation of mulberry accessions begins with their collection, multiplication, and subsequent establishment in ex-situ conservation sites. Different maintenance practices are essential to this process, including propagation methods, pruning techniques, and fertilisation strategies. The upcoming chapters will detail the maintenance methodologies employed by the germplasm collections of CREA, SCS Vratsa, and UM. These institutions serve as foundational models for other partners, providing a backbone for the effective conservation and maintainance of mulberry collections.
Methodology for mulberry germplasm characterisation and evaluation
Morphological traits of mulberry accessions maintained in the ARACNE partners’ collections (CREA, Vratsa, IMIDA, UM) were analyzed using UPOV Mulberry descriptors, basically modified according to the Guidance model to collect mulberry samples. To standardise the database of morphological descriptors across all partners, a template for an Excel file pivot table was developed. This template has been utilized by CREA, SCS Vratsa, IMIDA, and UM to ensure consistency in data recording and analysis. The list of descriptors is summarized in Table 2.
Table 2 - The list of mulberry descriptors employed for the evaluation of the mulberry genetic resources maintained in the germplasm collections of ARACNE partners.
|
Category |
Descriptor |
Options |
|
Taxonomical and basic information |
Identification/ Accession n. |
Data entry |
|
Species |
M. alba, M. indica, M. cathayana, M. celtidifolia, M. insignis, M. kordesiana, M. loboensis, M. macroura, M. mesozygia, M. microphylla, M. miyabeana, M. nigra, M. notabilis, M. rubra, M. serrata, M. trilobata, M. wittiorum, interspecific hybrid |
|
|
Varietal name |
Data entry |
|
|
Date of sampling |
Data entry |
|
|
Date of sampling |
Data entry |
|
|
Number of individuals in the repository |
Individual, Mulberry plantation, Mulberry row |
|
|
Method of observation |
Single measurement of a group of plants or part of plants, Measurement of a number of individual plants or parts of plants, Visual assessment by a single observation of group of plants or part of plants, Visual assessment by observation of individual plants |
|
|
Mode of origin/biological status |
Natural, Traditional cultivar/landrace, Advanced or improved cultivar, Hybrid |
|
|
Ploidy level (if known) |
Haploid, Diploid, Triploid, Tetraploid, Hexaploid, Docosaploid, Polyploid, Ploidy level not yet determined |
|
|
Geographical origin lat. |
Data entry |
|
|
Geographical origin long. |
Data entry |
|
|
Availability |
Public, Street, Square, Private Garden, Botanical Garden or Gene bank, Agricultural landscape |
|
|
Areas of cultivation |
Temperate, tropical, sub-tropical |
|
|
Morphological characteristics |
Trunk circumference (cm) |
<180 cm, 180-249 cm, 250-299 cm, >300 cm |
|
Exact measurement of trunk circumference (cm) |
Data entry |
|
|
Tree growth habit |
Upright, Semi-upright, Weeping |
|
|
Tree vigour |
Bad condition, Good condition |
|
|
Pruning practices |
Unpruned tree, Frequently pruned, Yearly pruned tree |
|
|
Trunk |
Trunk colour |
Greyish brown, Light brownish-grey, Reddish brown |
|
Trunk irregularities/damage |
Curved, Hollow (pipe tree), Longitudinally cracked, External split |
|
|
Shoots |
Current years shoots: length (cm) |
Data entry |
|
Current years shoot: number of lateral shoots |
Data entry |
|
|
Current years shoot: length of internode |
Data entry |
|
|
Colour of one-year old shoot |
Greyish brown, Greenish brown, Yellowish brown, Medium brown, Reddish brown, Dark brown |
|
|
Lenticel density |
High, Medium, Sparse |
|
|
Lenticel shape |
Elliptical, Oval, Round |
|
|
Buds |
Bud shape |
Broad triangular, Medium triangular, Narrow triangular, Ovate |
|
Bud colour |
Greyish brown, Yellowish brown, Reddish brown, Medium brown, Dark brown |
|
|
Bud size |
Small, medium, large |
|
|
Leaves |
Phyllotaxis |
Alternate spiral, Alternate distichous, Opposite decussate |
|
Leaf shape |
Simple, Lobed |
|
|
Peduncle length (cm) |
Data entry |
|
|
Average leaf size: length (cm) |
Data entry |
|
|
Average leaf size: width (cm) |
Data entry |
|
|
Average leaf size: area (cm2) |
Data entry |
|
|
Leaf blade |
Low (<1.2 broad leaves), Medium (1.3-1.5), High (>1.6 long) |
|
|
Petiole range |
Absent or very short (< 10 mm), Short (11-20 mm), Medium (21-40mm), Long (41-70 mm), Very long (>71 mm) |
|
|
Petiole size (cm) |
Data entry |
|
|
Shape of leaf base |
Cuneate, Truncate, Retuse, Cordate |
|
|
Shape of leaf apex |
Acute, Obtuse, Obcordate |
|
|
Leaf blade tip |
Absent, Caudate, Acuminate |
|
|
Leaf blade margin |
Repand, Crenate, Dentate, Serrulate, Biserrate, Serrate, Aristate |
|
|
Hairiness |
Glabrous, Midrib and veins, Evenly pubescent |
|
|
Glossiness |
Glossy, Matte |
|
|
Phenological descriptors |
Date of swollen bud (week) |
Data entry |
|
Date of bud burst (week) |
Data entry |
|
|
Flowering date (week) |
Data entry |
|
|
Date of first leaf (week) |
Data entry |
|
|
Date of first leaf (week) |
Data entry |
|
|
Early yellowing due to disease (week) |
Data entry |
|
|
Abundant autumn colouring (week) |
Data entry |
|
|
Leaf fall (week) |
Data entry |
|
|
Time of infructescence ripening |
Early, Early to medium, Medium, Medium to late, Late |
|
|
Uniformity of infructescence ripening |
Early, Early to medium, Medium, Medium to late, Late |
|
|
Reproductive structures |
Sexual dimorphism |
Monoecious, Dioecious |
|
Inflorescence type |
Male, Female, Predominantly female with some male flowers at the base, Predominantly male with some female flowers at the apex, Predominantly male with some female flowers at the base, Predominantly female with some male flowers at the apex |
|
|
Inflorescence size |
Female inflorescence length (mm) |
Data entry |
|
Female inflorescence diameter (mm) |
Data entry |
|
|
Female inflorescence stalk length (mm) |
Data entry |
|
|
Male inflorescence length (mm) |
Data entry |
|
|
Male inflorescence length (mm) |
Data entry |
|
|
Male inflorescence stalk length (mm) |
Data entry |
|
|
Polygamous inflor. length (mm) |
Data entry |
|
|
Polygamous inflor. diameter (mm) |
Data entry |
|
|
Polygamous inflor. diameter (mm) |
Data entry |
|
|
Infructescence |
Fruit weight (g) |
Data entry |
|
Length (mm) |
Data entry |
|
|
Width (mm) |
Data entry |
|
|
Length of peduncle |
Short, Medium, Long |
|
|
Colour |
Yellowish white, Light pink, Purple brown, Reddish black, Black |
|
|
Taste |
Acidic, Sweet, Balanced |
|
|
Infructescence shape |
Ovoid/globose, Ellipsoid, Cylindrical, Irregular |
|
|
Growth and yield attributes |
Growth |
Slow, Medium, Fast |
|
Length of primary branch (cm) |
Data entry |
|
|
Internodal distance (cm) |
Data entry |
|
|
Leaf No./meter |
Data entry |
|
|
Weight of 100 fresh leaves (g) |
Data entry |
|
|
Chemical composition of leaves |
Total protein (g/100 g) |
Data entry |
|
Mineral (g/100 g) |
Data entry |
|
|
Fibre (g/100 g) |
Data entry |
|
|
Response to different physological conditions |
Response to drought |
Resistant, Tolerant, Susceptible |
|
Response to salinity |
Resistant, Tolerant, Susceptible |
|
|
Response to chilling stress |
Good, Medium, Poor |
|
|
Response to repeated pruning |
Good, Medium, Poor |
|
|
Diseases and pests |
Disease option |
Fungal leaf spot, Bacterial leaf spot (Pseudomonas syringae pv. mori), Soft rot (Pectobacterium carotovorum), Ringspot virus |
|
Pest option |
Mulberry moth (Hyphantria cunea), Thrips (5 species), Mites (Tetranychus sp.), Scale insects (Hemiptera), Mealy bugs (Maconellicoccus hirsutus), Red scale (Aonidella aurantii), Hairy caterpillar (Spilarctia obliqua), Jassids (Empoasca Flavescens) |
|
|
Any other disease or pest |
Data entry |
|
|
Leaf necrotic spots |
Low, medium, frequent |
|
|
Bark lesions |
Frequent, Few |
|
|
Additional remarks |
Optional |
Data entry |
For the evaluation of morphological descriptors, varieties were meticulously selected from each germplasm collection to ensure a comprehensive and comparable analysis. The main morphological features were studied during the years 2023-2025. Supporting schematic and pictorial information of the studied descriptors are shown in Figure 1-5.

Figure 1: Morphological characterization of the one-year old shoots and buds. A) Colour of the one year old shoot, B) Bud colour, C) bud shape, D) Lenticel shape.
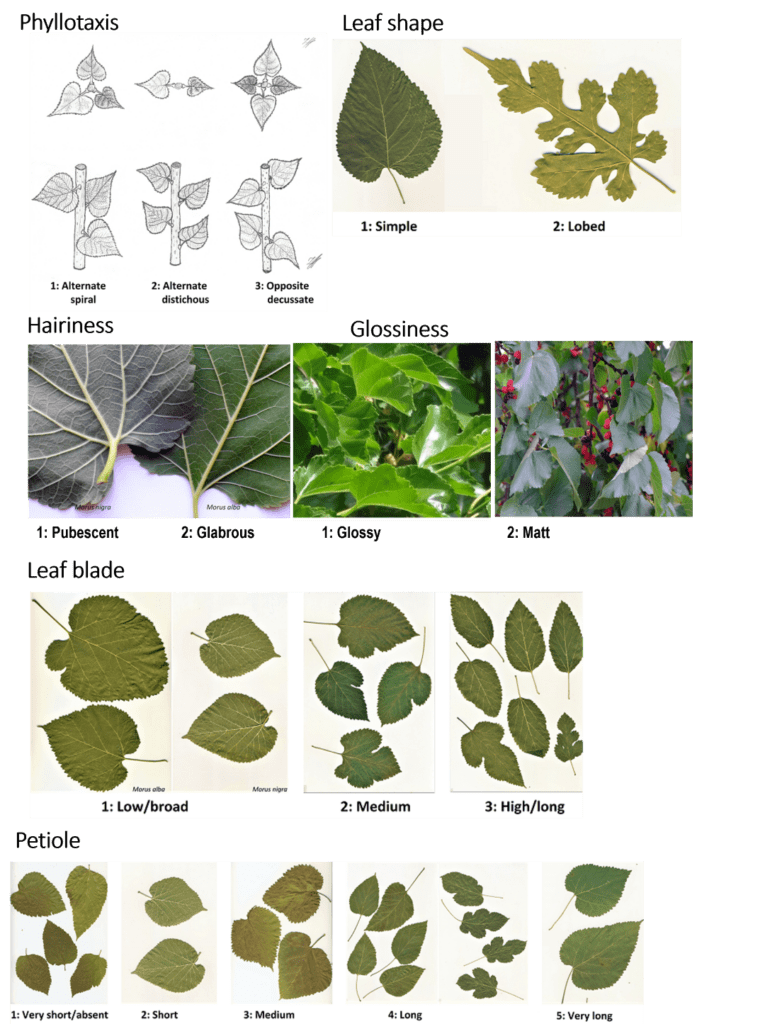
Figure 2: Morphological characterization of the leaves. Phyllotaxis, leaf shape, hairiness, glossiness, leaf blade, petiole.
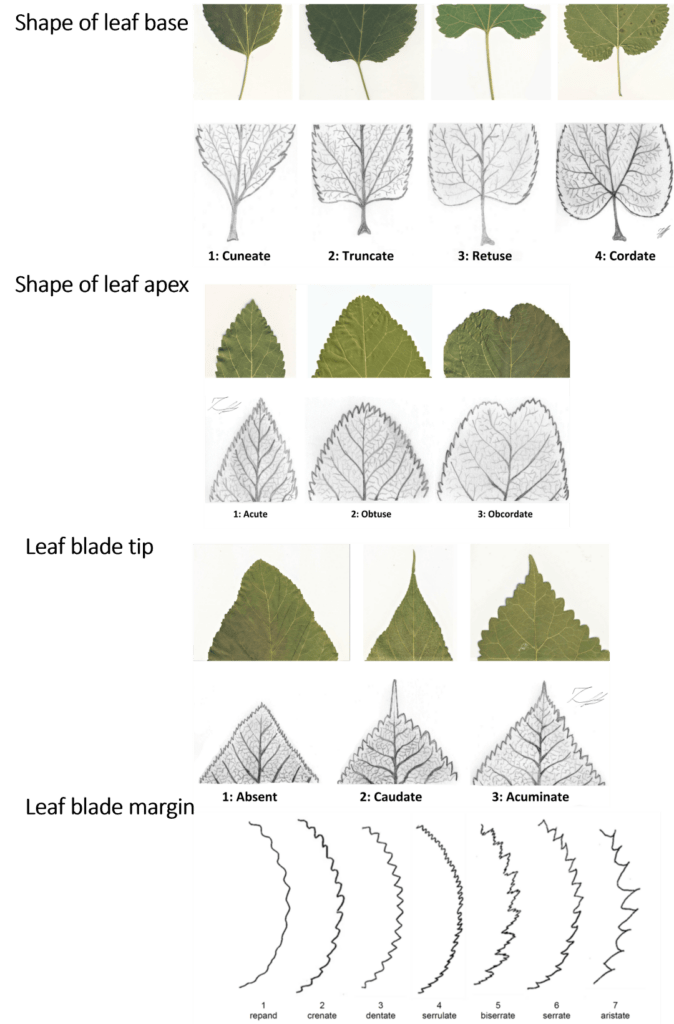
Figure 3: Morphological characterization of the leaves. Shape of leaf base, shape of leaf apex, leaf blade tip, leaf blade margin.

Figure 4: Morphological characterization of the reproductive structures. Inflorescence types, stigma persistency length of infructescence peduncle, uniformity of infructescence ripness.

Figure 5: Morphological characterization of the reproductive structures. Colour of infructescence, infructescence shape.

References
1. Ahlawat, T., Patel, N. L., Agnihotri, R., Patel, C. R., & Tandel, Y. (2016). Black mulberry (Morus nigra). Underutilized fruit crops: Importance and cultivation, 195-212.
2. Alberti J. (1773) “Dell’epidemica mortalità de’ gelsi e della cura e coltivazione loro”. Ed. Bartolommeo Richitti, Salo’.
3. Battistini, F. (2003). L’industria della seta in Italia nell’età moderna. Il Mulino, Bologna, 257 p.
4. Benedetti M. & Fila G. (2023). Biodiversità agraria da salvare: i gelsi secolari di Vivaro. Vita in Campagna 4, 56-59.
5. Bolle, I. (1896). Poduk o Murvoreji. Available online: https://www.dlib.si/stream/URN:NBN:SI:DOC-ICEMEK1Z/ad0fff95-e4c9-4763-a02d-70a28296f89c/PDF
6. Bolle, I. (1908). Ob Uzgoju Murava. Available online: https://www.dlib.si/stream/URN:NBN:SI:DOC-QCY0RSOI/6906e8d3-fb09-4249-89b5-fcea5f27a09e/PDF
7. Browicz, K. (2000). Where is the place of origin of Morus nigra (Moraceae)?. Fragmenta Floristica et Geobotanica, 45(1/2), 273-280.
8. Brus, R., & Surina, B. (2012). Drevesa in grmi Jadrana. Modrijan.
9. Burlando, B., Clericuzio, M., & Cornara, L. (2017). Moraceae plants with tyrosinase inhibitory activity: A review. Mini reviews in medicinal chemistry, 17(2), 108-121.
10. Cappellozza, L. (2000). Present situation of mulberry germplasm resources in Italy and related projects. In Electronic conference on Mulberry for Animal Production (Vol. 1).
11. Cappellozza, L. (2002). (https://www.fao.org/3/AD107E/ad107e19.htm, access: 10.11.2023)
12. Carpenè, A., & Vianello, A. (1874). La vite e il vino in provincia di Treviso. Reproduced from E. Loescher press, Torino, by De Bastiani press, Vittorio V. (TV), 186 p.
13. Chang, S., Wu, C., & Cao, Z. (1998). Moraceae. Flora Reipublicae Popularis Sinicae, 23(1), 1-245.
14. Clement, W. L., & Weiblen, G. D. (2009). Morphological evolution in the mulberry family (Moraceae). Systematic Botany, 34(3), 530-552.
15. Coles, P. (2022). The wrong mulberries? Moriculture before the white mulberry. Textiles and Clothing Along the Silk Roads: Thematic Collection of the Cultural Exchanges along the Silk Roads, 1, 117.
16. CREA Planta res, (2013). (http://planta-res.politicheagricole.it/pages/index.php, access: 02.12.2023).
17. Durand, A., Bouby, L., Chabal, L., Mane, P., & Ruas, M. P. (2016). Histoire et utilisation des mûriers blanc et noir en France. Apport de l’archéobotanique, des textes et de l’iconographie.
18. Gelorini, V., & Bourgeois, J. (2005). First Discovery of Black Mulberry (Morus nigra L.) Pollen in a Late Bronze Age Well at Sint-Gillis-Waas (Flanders, Belgium): Contamination or in situ Deposition?. Environmental Archaeology, 10(1), 91-96.
19. Ghosh, A., Gangopadhyay, D., & Chowdhury, T. (2017). Economical and environmental importance of mulberry: a review. International Journal of Plant and Environment, 3(2), 51-58.
20. Hlubek, F.X. (1850). Unterricht in der Maulbeerbaum-und Seidenzucht für das Landvolk; Steiermärkische Seidenbau-Vereine: Graz, Austria.
21. Hotta, T. (1954). Fundamentals of Morus plants classification. Kinugasa Sanpo, 390, 13-21.
22. Hou, A. J. (2015). The Moraceae family: an important source of drug discovery and development. Planta Medica, 81(05), OA6.
23. International Plant Names Index (2024) available online: https://www.ipni.org/?q=morus, accessed online: 04.01.2024
24. Jian, Q., Ningjia, H., Yong, W., & Zhonghuai, X. (2012). Ecological issues of mulberry and sustainable development. Journal of resources and ecology, 3(4), 330-339.
25. Jiang, Y., Huang, R., Yan, X., Jia, C., Jiang, S., & Long, T. (2017). Mulberry for environmental protection. Pakistan Journal of Botany, 49(2), 781-788.
26. Kew (2024) (available online: https://powo.science.kew.org/taxon/, accessed: 10.01.2024).
27. Liu, Y., & Willison, J. M. (2013). Prospects for cultivating white mulberry (Morus alba) in the drawdown zone of the Three Gorges Reservoir, China. Environmental Science and Pollution Research, 20, 7142-7151.
28. Macflot, M., (1776), Vollständige aus Vieljähriger Erfahrung Vollkommen Gegründete Anleitung Sowohl zur Seidenzucht als Auch Zum Pflanzen und Beschneiden der Maulbeerbäume; Hansebooks: Norderstedt, Germany, p. 84.
29. Maguire, T. L., Collins, G. G., & Sedgley, M. (1994). A modified CTAB DNA extraction procedure for plants belonging to the family Proteaceae. Plant Molecular Biology Reporter, 12, 106-109.
30. Masera, E. (1968). Contributo del Baco da Seta al Progresso Umano. Italian Journal of Zoology, 35(4), 493-505.
31. MorusApp (2023), available online: https://morusapp.aracneproject.eu/login, accessed online: 14.11.2023
32. Nepal, M. P. (2008). Systematics and reproductive biology of the genus Morus L.(Moraceae). Kansas State University.
33. Nepal, M. P., & Purintun, J. M. (2021). Systematics of the Genus Morus L.(Moraceae): Taxonomy, Phylogeny and Potential Responses to Climate Change. In Mulberry: Genetic Improvement in Context of Climate Change (pp. 2-20). CRC Press.
34. Pliny the Elder. (77 AD). Naturalis Historia, book XVI, cap LXI, v 102
35. Powell W., Morgante M., Andre C., Hanafey M., Vogel J., Tingey S. and Rafalski A. (1996). The comparison of RFLP, RAPD, AFLP and SSR (microsatellite) markers for germplasm analysis. Molecular Breeding 2: 225–238
36. Rohela, G. K., Shukla, P., Kumar, R., & Chowdhury, S. R. (2020). Mulberry (Morus spp.): An ideal plant for sustainable development. Trees, Forests and People, 2, 100011.
37. Rudolf (Kronprinz von Österreich und Ungarn). (1891). Die österreichisch-ungarische Monarchie in Wort und Bild: Das Küstenland (Görz, Gradiska, Triest und Istrien), Vienna: Wien K.K. Hof- und Staatsdruckerei, p. 287.
38. Silbermann, H. (1870). Die Seide, ihre Geschichte, Gewinnung und Verarbeitung… .
39. The Fruit of Broken Dreams (2000), available online: https://www.latimes.com/archives/la-xpm-2000-jul-19-fo-55036-story.html, accessed online: 15.01.2024
40. The Mulberry Tree (2016), available online: https://charltonsoc.files.wordpress.com/2021/02/the-mulberry-tree_peter_coles.pdf, accessed online: 15.01.2024
41. Wimmer 1842. Theophrastus – https://books.google.si/books?id=110GAAAAQAAJ&hl=sl&source=gbs_navlinks_s
42. Theophrastus. (1552). Theophrasti … De historia plantarum :libri IX cum decimi principio, & De causis, siue, earum generatione, libri VI /Theodoro Gaza interprete.
43. Tikader, A., & Vijayan, K. (2017). Mulberry (Morus spp.) genetic diversity, conservation and management. Biodiversity and conservation of woody plants, 95-127.
44. Time line of the mulberry in London (2016), available online: https://www.moruslondinium.org/research/timeline, accessed online: 15.01.2024
45. Trigueros, R. O., & Villalta, P. (1997). Evaluación del uso de follaje deshidratado de morera (Morus alba) en alimentación de cerdos de la raza Landrace en etapa de engorde. San SV, 150-155.
46. Urbanek Krajnc, A., Bakonyi, T., Ando, I., Kurucz, E., Solymosi, N., Pongrac, P., & Berčič, R. L. (2022). The Effect of Feeding with Central European Local Mulberry Genotypes on the Development and Health Status of Silkworms and Quality Parameters of Raw Silk. Insects, 13(9), 836.
47. Urbanek Krajnc, A., Senekovič, J., Cappellozza, S., & Mikulic-Petkovsek, M. (2023). The Darker the Better: Identification of Chemotype Profile in Soroses of Local and Introduced Mulberry Varieties with Respect to the Colour Type. Foods, 12(21), 3985.
48. Urbanek Krajnc, A., Ugulin, T., Pausic, A., Rabensteiner, J., Bukovac, V., Mikulic Petkovsek, M., … & Felicijan, M. (2019). Morphometric and biochemical screening of old mulberry trees (Morus alba L.) in the former sericulture region of Slovenia. Acta Societatis Botanicorum Poloniae, 88(1).
49. Urbanek Krajnc, Andreja & Rabensteiner, Johannes. (2018). Geschichte über Grenzen hinweg Maulbeerbäume als lebende Monumente : der Fast vergessenen Seidenkultur = Zgodba, ki sega čez meje murve – živi spomeniki : skoraj pozabljene kulture gojenja sviloprejk. VII : das Magazin der Sloweninnen und Slowenen in der Steiermark.
50. Urbanek Krajnc, A., Senekovič, J., Jelen, Š., Kozmos, M., Rabensteiner, J., Mikulič Petkovšek, M., Osterc, G. (2024). Phytochemical characterisation of the soroses of the old local mulberry genotypes and reintroduction of moriculture in Slovenia with the aim of different uses in agriculture = Fitokemična karakterizacija plodov starih lokalnih genotipov murv in ponovno uvajanje pridelave murv v Sloveniji z namenom vsestranske uporabnosti v kmetijstvu. V: Hudina, M.(ur.). Zbornik referatov 5. slovenskega sadjarskega kongresa z mednarodno udeležbo : Krško,. 1. elektronska izd. Ljubljana: Strokovno sadjarsko društvo Slovenije, . Str. 177-189, ISBN 978-961-91301-6-2
51. Verson E. & Quajat E. (1896) “Il filugello e l’arte sericola”. Ed. F.lli Drucker, Padova-Verona.
52. Vijayan, K., Gnanesh, B. N., & Tikader, A. (2023). Botanical Features and Economic Significance of Mulberry. In The Mulberry Genome (pp. 1-11). Cham: Springer International Publishing.
53. Vijayan, K., Tikader, A., Weiguo, Z., Nair, C. V., Ercisli, S., & Tsou, C. H. (2011). Morus. Wild crop relatives: genomic and breeding resources: tropical and subtropical fruits, 75-95.
54. Wani, S. A., Bhat, M. A., Malik, G. N., Zaki, F. A., Mir, M. R., Wani, N., & Bhat, K. M. (2013). Genetic diversity and relationship assessment among mulberry (Morus spp) genotypes by simple sequence repeat (SSR) marker profile. African Journal of Biotechnology, 12(21).
55. WFO Plant list (2024), available online: https://wfoplantlist.org/taxon, accessed online: 04.01.2024
56. Yamanouchi, H., Koyama, A., & Machii, H. (2017). Nuclear DNA amounts of mulberries (Morus spp.) and related species. Japan Agricultural Research Quarterly: JARQ, 51(4), 299-307.
57. Yang, C. X., Liu, S. Y., Zerega, N. J., Stull, G. W., Gardner, E. M., Tian, Q., … & Yi, T. S. (2023). Phylogeny and biogeography of Morus (Moraceae). Agronomy, 13(8), 2021.
58. Yang, J., Chu, Q., Meng, G., & Kong, W. (2022). The complete chloroplast genome sequences of three Broussonetia species and comparative analysis within the Moraceae. PeerJ, 10, e14293.
59. Zeng, Q., Chen, H., Zhang, C., Han, M., Li, T., Qi, X., … & He, N. (2015). Definition of eight mulberry species in the genus Morus by internal transcribed spacer-based phylogeny. PloS one, 10(8), e0135411.
60. Zeng, Q., Chen, M., Wang, S., Xu, X., Li, T., Xiang, Z., & He, N. (2022). Comparative and phylogenetic analyses of the chloroplast genome reveal the taxonomy of the Morus genus. Frontiers in Plant Science, 13, 1047592.
61. Zimmermann, P. (2016). Štajerski raj: Savinjska dolina in Novo Celje. Žalec: ZKŠT – Zavod za kulturo, šport in turizem.
